- Future Electronics Singapore Celebrates International Women's Day 2024
- Infographic informs about Chicago Medical Malpractice Lawyer
- Indiatimes 2.0 redefines digital storytelling for modern youth
- Weinberg Law Offices Highlights the Vital Role of a Los Angeles Lawyer in Dog Bite Cases
- Future Electronics Contributes to Reforestation Efforts in Quebec for Earth Day 2024
- The Yoga Institute Announces Free Access to "Samattvam" Yoga OPD in Honor of Dr. Jayadeva Yogendra's Birth Anniversary
- Over 900 Exhibitors to Showcase Food & Beverage Innovation to More Than 27,000 Visitors at JFEX 2024
- Ellen Christman and Nicole Guidi to Launch Discovery Map of Sarasota
- Sennheiser unveils the latest generation flagship earbuds with the launch of MOMENTUM True Wireless 4 in India
- University of Sunderland Delegation Visits Kings Cornerstone International College to Meet Future Transfer Students
- Paintings by Maud Lewis Lead The Way in Miller & Miller's Successful Online Auction Held April 13th
- GD Goenka University Inaugurates State-Of-The-Art Health & Wellness Centre In Association With Park Hospital
- Betachon Freight Auditing Announces Exclusive Discount Ocean Freight Rates with DHL International Shipping
- Nourhan Rabah Bureau Redefines Urban Living in Dubai
- TiDB by PingCAP Leads Data Management Revolution at Great International Developer Summit 2024
 Mail to a Friend Mail to a Friend |
|
     |
Akcea and Ionis Announce Publication of Long-Term Clinical Data of TEGSEDI® in Patients with Polyneuropathy Driven by hATTR Amyloidosis
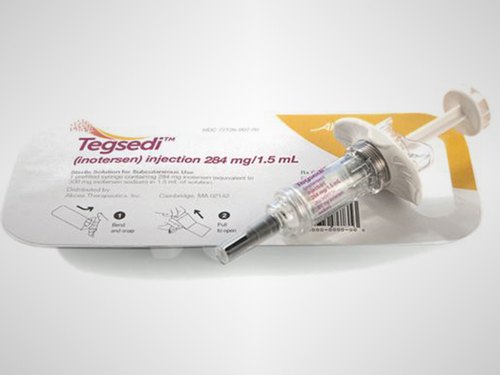
Results from the ongoing, open-label extension (OLE) study of the pivotal NEURO-TTR trial published in the European Journal of Neurology show that patients treated with TEGSEDI? (inotersen) experienced sustained improvements in measures of neuropathic progression at 39 months of therapy compared to the natural history of the disease.
Patients administered TEGSEDI also experienced clinically relevant improvements in measures of quality of life compared to the natural history of the disease.
No new safety signals were identified in the OLE study.
Company :-Akcea Therapeutics Inc.
User :- Akcea Therapeutics
Email :-info.akceacanada@gmail.com
Phone :-(617) 207-0202
Url :- http://www.akceatx.ca/media/press-releases/











