- Media and Entertainment Skill Council to Expand Its Initiatives
- Sociapa Bags the Digital and Creative Mandate for Madhusudan Dairy
- Vancouver Interior Painting Now Offering Complete Interior Painting and Drywall Repair Services in Greater Vancouver
- VMS General Trading LLC-Announces Unbeatable Bulk A4 Paper Sale
- Karan Johar and Guneet Monga Kapoor’s \'Gyaarah Gyaarah\' trailer breaks time barriers on ZEE5
- JIS Fall Registration Now Open: Showcasing Fall’s Finest Trends and a Vibrant Buying Experience
- The Creative Leather Unveils Iconic Leather Trench Coat for the Modern Trendsetter
- Israel Mgdesyan, Head of the sales department of AVTODOM Electro, spoke about the features of operating LiXiang electric cars
- Samurai Movers Expands Exceptional Moving Services to Westminster and Wheat Ridge
- Meticulous Research® Publishes New Report on the Li-ion Battery Recycling Market
- Studio52 Listed Among the Top Animation Production Companies in the UAE
- The Animal Care Organization Inaugurates a Veterinary Hospital with Animal Birth Control Unit & Mobile Health Van at Faridabad
- Barcelona Premier Soccer Club Announces the Launch of Elite Soccer Academy in San Antonio
- Xedos Technologies Announces Launch of Xedos Digital, a Comprehensive Digital Marketing Division
- STEMart Announces Transportation and Distribution Simulation Services for Medical Device Manufacturers
 Mail to a Friend Mail to a Friend |
|
   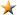  |
Hester Biosciences Ltd reports Net Profit of Rs. 3.56 crore and Revenues from Operations of Rs. 50.7 crore in Q1FY23
Ahmedabad: 10 August 2022: One of India\'s leading poultry and animal vaccine manufacturing company, Hester Biosciences Limited has reported consolidated net profit of Rs. 3.56 crore and Revenue from Operations of Rs. 50.70 crore for the Q1FY23. Individually, the gross margins of the vaccines and the health products have been in line with the corresponding quarter, however, the overall margins have reduced due to the increase in the proportion of health products sales which have lower gross margins compared to vaccines. Health Products sales constituted 34% of the total sales in Q1 FY23, versus 20% in Q1 FY22.
Hester Tanzania has recently received regulatory approvals for four products with two additional products under approval process. Hester Tanzania has just started commercial operations and Revenues are expected to start in Q2. Hester Nepal has registered 16% growth in domestic revenues, however, there were no tender sales in the current quarter due to delay in tendering by FAO and other multilateral institutions.
Consolidated Financial Highlights
Operating EBITDA and PAT margin have declined due to :
(a) Reduction in vaccine sales as explained above
(b) Relative increase in marketing expenses in Q1 FY23 as against in Q1 FY22 which were significantly lower due to Covid travel restrictions (notwithstanding the higher sales in Q1 FY22 due to disease situation in poultry)
(c) Increased market development cost in Animal Health and Petcare Divisions on account of company?s continuous efforts in expansion activities. These are expected to moderate over next few quarters as growth in sales picks up in new territories.
Going further - The company is confident of arresting the degrowth in Q2 as well as hopes to improve the profitability as follows:
On the vaccine side:
1. The recent outbreak of Lumpy Skin Disease (LSD) in certain parts of the country since July 2022 is expected to result in additional sales in Q2 FY23.
2. The Company was a successful bidder in a government tender for PPR vaccine for sheep & goat, the supplies for which are likely to start from September 2022.
On the Health Product side:
It is our endeavour to improve the profitability. Over next couple of quarters, Hester will focus on growing sales on back of improving the sales productivity of the marketing team as well as launching new products and entering new territories.
Petcare division
The Company launched a new division for Petcare during Q1 with 10 products. Activities related to market development, field force establishment and product pipeline are ongoing. Petcare Division will emerge as a steady long term growth driver, given the increasing adoption of pets in the country.
Status on Hester?s initiatives in Covid-19 vaccine
? The Company, in consortium with Gujarat Biotechnology Research Centre (GBRC), Government of Gujarat (GoG), has entered into a term sheet agreement with Bharat Biotech India Limited to manufacture the Drug Substance for Covaxinunder the Mission Covid Suraksha Scheme of Government of India.
? The project is to manufacture Drug Substance equivalent of up to 7 million doses per month.
? The construction of BSL-3 facility is nearing mechanical completion.Planning andpreparation for commissioning is ongoing. The facility is expected to be ready for commercial operation in Q3FY23.
? This facility is a multi-purpose facility to handle other micro-organisms beyond Covid-19.
Other developments
1. The Company is strengthening its new product vaccine pipeline by developing new vaccines like Classical Swine Fever (CSF), Sheep Pox and an improved version of Brucella vaccines.
2. Bulk antigen production capacity expansion project is completed and trial runs are ongoing. Expansion of Fill-Finish line capacity is expected to be completed by Q4FY23. These 2 expansions will double the production capacity in vaccines.
3. The recent notification by Government of India to allow the manufacture and sale of Avian Influenza Inactivated vaccine, H9N2 strain, will contribute to our sales from Q3.
User :- Gopal Modi
Email :-gopal@coldcococonsultancy.com










