- Synology Introduces Advanced Data Management and Security Solutions Against the Rising Ransomware Threats in India
- Shannon's Spring Fine Art Auction on Thursday, May 2nd, will be Led by Important American Paintings
- RITES Limited DVC collaborate for rail infra works
- Holabird's Wild West Relics Auction, April 6-7 & 13-14, had 2,300 Lots in 60 Collecting Categories
- Extraordinary People Awards and Williams Elite Hall of Honor Embrace International Guest for 2024
- Mapic India Shopping Centre and Retail Summit & Awards 2024 slated to happen in Delhi on April 25th
- Toradex Strengthens Custom Solutions Offering with Acquisition of Linear Computing Inc.
- ATC Income Tax Unveils Powerful EITC Calculator to Maximize Tax Returns for Eligible Taxpayers
- Webskitters Academy Introduces Advanced Blockchain Developer Course
- Southshore Dental Launches New Website Dedicated to Dental Implants
- Housing Guards Announces Expanded Inventory of Drywall Supplies in Uxbridge
- BML Munjal University institutes School of Liberal Studies Conversations organizes the Inaugural Edition on Hindi: Vaad-Vivaad-Samvaad
- Yashoda Hospitals, Hyderabad, Successfully Treats a 60-year-old from Bangladesh for Carcinoma Lung
- Creative Biolabs Introduces Innovative Antibody-Drug Conjugate Development Services
- Documentationconsultancy.com is Ready to Sell the CMMI V3.0 Documents Online for IT Industries
 Mail to a Friend Mail to a Friend |
|
   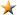  |
Sepha hits one micron target with new HD leak tester

Sepha has achieved micro leak detection down to one micron (1?m) with the development of a new high performance leak tester for CCIT (Container Closure Integrity Testing) applications. Utilising the vacuum decay test method, the new Multi-Q HD unit takes integrity testing to the next level, enabling ultra-sensitive, non-destructive detection of defects in pharmaceutical containers. This enhanced testing functionality responds to demand from parenteral formats, which are experiencing unprecedented growth on the back of international Covid-19 vaccination programmes.
Explaining the context to the development of the new Multi-Q HD, Sepha?s Managing Director, Paul Smith, says: ?Testing to 5?m used to be the benchmark for CCIT, but in recent years, we have witnessed a drive from the industry to raise the bar for increased integrity test sensitivity. Since the pandemic, this trend has really gained momentum.?
He continues: ?Intercepting defective containers before they reach end-users or patients is important for any pharmaceutical, but for short shelf life, highly sensitive, injectable products it is absolutely crucial. Ingress of air through micro cracks and pinholes has implications for product efficacy and safety, therefore pharmaceutical manufacturers cannot afford to be anything less than 100% confident in the integrity of their parenteral formats. We view it as our responsibility to provide the technology that can instil this level of confidence. With this in mind, we set ourselves the target of guaranteeing micro leak detection down to 1?m and we are proud and delighted to report that we have achieved that goal.?
Sepha?s engineers have achieved this step-change in detection performance with an advanced sensor configuration that filters out interference.
?With a standard sensor, there is a certain level of signal-to-noise ratio that makes it difficult to pick up the slight rise in pressure that is usually indicative of a micro leak. At the heart of the Multi-Q HD unit is sensor technology with an improved signal-to-noise ratio, so that even the most minuscule pressure differential is discernible. The sensor is also located at the point of measurement, which yields a faster response and more sensitive results compared to alternative technologies,? explains Smith.
Building on Sepha?s established Multi-Q leak testing platform, the Sepha Multi-Q HD is a standalone, lab-scale leak test system that can be used in combination with interchangeable attachments to fit the full range of parenteral container types available on the market today. These include glass or plastic vials, ampoules, pre-filled syringes, bottles and BFS (Blow Fill Seal) containers. Its compact, bench-top design means it can be used in the lab during drug development or for in-process batch testing to verify the integrity of containers being manufactured on the production line.
Whilst the Multi-Q HD can be configured to perform a variety of non-destructive, deterministic tests, such as pressure decay, Sepha expects that it will primarily be used for applications which require a vacuum decay test. The Multi-Q HD allows manufacturers to test in accordance with the ASTM Standard Test Method F2338-09 for vacuum decay in line with FDA consensus and USP 1207 guidelines. This non-destructive and deterministic test method provides quick, reliable and repeatable results without any damage to the product or packaging.
Another advantage of the Multi-Q HD is that it incorporates as standard, a number of features that are often only available as add-ons with other manufacturers. These include OPC connectivity, Active Directory, 21 CFR Part 11, Electronic Signatures and unlimited data storage.
The new Multi-Q HD will be launched via a live webinar on 23rd June 2021 where Sepha?s packaging inspection experts will lead an online demonstration of the system.
Webinar details
Wednesday 23rd June: 09.00 UTC+1
Wednesday 23rd June: 14.00 UTC+1
https://app.livestorm.co/sepha/reliable-ccit-down-to-1-micron
The webinar will also be available on demand following the live event.
Company :-Scott PR
User :- Lucy Shaw
Email :-lrs@scottpr.com
Phone :-01477898745
Url :- https://sepha.com









